All you need to know about CBD

What is CBD?
CBD is a chemical that is commonly extracted from the hemp plant and its cousin the marijuana plant. While CBD is an active ingredient of cannabis, it does not cause users to experience a “high” or psychoactive effect like tetrahydrocannabinol (THC) – another chemical found in the plants.
The CBD found in food is usually taken from hemp oil, which contains less THC than that found in marijuana. While tests by the NHS are inconclusive, some studies have claimed CBD can be used for pain relief, easing anxiety and – in some cases – help to battle epilepsy and multiple sclerosis.
The potential presence of contaminating THC has associated implications under the Misuse of Drugs Act 1971. As a result, many experts are currently counselling caution, avoiding any effective presence of THC in foods containing CBD.
The growth in the CBD edibles market
The global CBD market is estimated to grow to US$2.17bn by 2023, up from $462m reported in 2018.
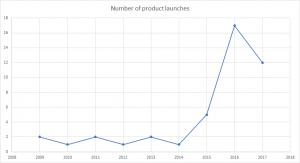
Europe has become a hotbed of hemp-based product launches in recent years. A report from Persistence Market Research found that there had been 256 new food and drink products launched across the region from 2008–2017, almost double the number of launches in the US.
Persistence senior consultant Pratik Gurnani said: “A wide range of hemp-based edible products are available in many stores across the UK. CBD gummies, CBD soft gel capsules, ready-to-drink CBD beverages, as well as flavoured CBD tea and coffee are some of the popular products across the UK.
“The rapidly growing CBD market – for food supplements and cosmetics – hit €144m (£133m) in 2017 and is projected to reach €850m (£787m) by 2020.”
CBD product launches in the UK
Legal challenges surrounding CBD
Under EU law, CBD-extract products are considered novel foods. As such, manufacturers producing food containing CBD in the UK need to comply with the novel foods process and submit safety information to the European Food Safety Authority in support of their products. As of writing, no manufacturers have submitted their products for review, according to the Food Standards Agency.
A spokesman for Leatherhead Food Research added: “Any extracts from the Cannabis sativa plant, created by other industrial processes than cold pressing used to produce Cannabis sativa L, seed oil, and any derived products containing CBD, are considered as novel in the EU, and will therefore have to be submitted for a pre-market safety assessment under the EU Novel Food legislation.
“The Novel Food application process is crucial in ensuring that any food or food ingredients not available on the EU market before 15 May 1997 are safe before reaching consumers. Due to the complexities associated with the cannabis and hemp species, varieties, extraction methods, purification methods and so forth, the journey is not expected to be smooth sailing.”
CBD drinks in the UK
While some larger food and drink companies have shown an interest in CBD in the past, the realm is being dominated by start-ups in the sector. Much like the non-alcoholic spirits and wine sector, smaller players are leading the charge into the unknown territory of CBD.
Companies like Relax Brands-owned Drink 420 are promoting CBD-infused drinks as an all-natural alternative to sugary soft drinks and are promoting the high levels of CBD in the drinks on the packaging.
“We provide an amazing, top-quality product with a great amount of CBD inside, 15mg, which is the highest on the UK market, because we know consumers are concerned with the efficacy of CBD products,” said Relax.
“We always third party-test our products to ensure the level of CBD stated is actually what is inside – something over which brands in the USA have recently come into disrepute.
“The CBD market as a whole is growing exponentially thanks to growing awareness and market saturation. Based on our research, this is only the start of CBD-infused beverages, as consumers are looking to take CBD in a convenient, inclusive way.”
Issues surrounding export to the US
CBD currently sits in a grey area in the US, which makes the prospect of exporting food and drink containing the ingredient to the country difficult.
Confusion began in December last year, when US President Donald Trump signed the $867bn Farm Bill. Part of this bill saw industrial hemp removed from the US Government’s list of controlled substances, making it legal for companies to make good from the product – including food and drink.
However, the US Food and Drug Administration (FDA) still prohibits firms from adding CBD to food and drink, a chemical found within the hemp-based products that have now been legalised.
While there has been a boom in CBD consumption in the US, the FDA is keen to crack down on it, citing safety concerns. As such, the organisation is looking for ways to legalise the sale of CBD oil in food and drink, but such a move would require it to outline manufacturing conditions for the chemical.